Ivermectin and COVID-19 Review: Updated February 2021
As of March 2021, there are 50 on-going trials on Ivermectin and COVID-19 registered on ClinicalTrials.gov.
Ivermectin is an anti-parasitic medication widely used in low- and middle-income countries to treat
parasitic worm infections in adults and children. It’s been used for decades for this purpose by over 3.7
billion people, and is considered safe and effective. It has an increasing list of indications due to
its antiviral and anti-inflammatory properties, and is included on the WHO’s Model List of Essential
Medicines.
Ivermectin and COVID-19 Updates:
Feb 25, 2021: Drug used to treat lice and scabies drug could cut Covid deaths by up to 75%, research suggests, DailyMail reported.
Jan 23, 2021: Researchers at Oxford University are planning the first, large high-quality trial of Ivermectin that has been credited with dramatically reducing Covid-19 deaths in the developing world. The PRINCIPLE trial is aiming to find a drug that works soon after virus symptoms appear in a patient, and one that is most effective during the primary stages of the illness, The Times reported.
Jan 19, 2021: A pilot study published in the Lancet on January 19, 2021 showed some promising results but the authors concluded that the study warrants further exploration under larger trials with clinical outcomes in patients with risk factors or more severe disease.
Jan 14, 2021: The National Institutes (NIH) has issued a new statement on the use of the anti-parasitic drug ivermectin for the treatment of COVID-19. Previously, it recommended against this treatment, but now states that its Panel “has determined that there are insufficient data to recommend either for or against the use of ivermectin for the treatment of COVID-19.”
Jan 13, 2021: The BIRD meeting (British Ivermectin Recommendation Development) was convened by Dr. Tess Lawrie in order to present the findings from her rapid systematic review and meta-analysis of studies on the use of ivermectin to prevent and treat COVID-19. Dr. Lawrie presented evidence in the form of a DECIDE evidence-to-decision framework, a format used by the World Health Organization for the development of guidelines and recommendations in medical practice. Twenty experts from around the world and the UK attended the meeting, including 13 clinicians, and seven representatives from the public.
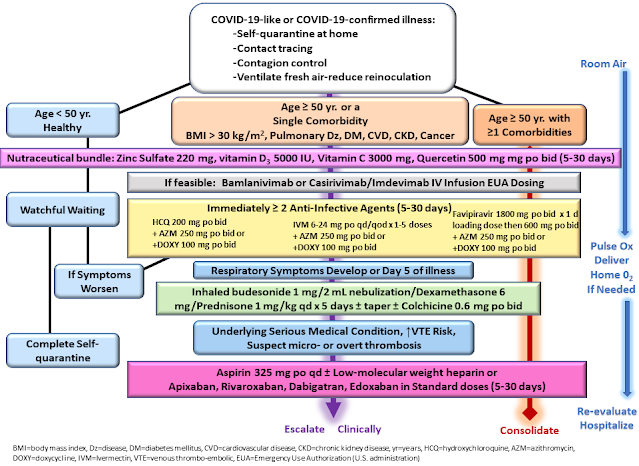
Dec 30, 2020: An essential updated review of COVID-19 early-treatment best practices was published. (abstract | PDF | HTML)
This international collaboration — comprised of physicians, like lead author Peter McCullough, MD, courageously treating patients despite the prevalence of “therapeutic nihilism” among government agencies like the NIH and FDA — outlines the urgency of, “prompt early initiation of sequenced multidrug therapy (SMDT) … to stem the tide of hospitalizations and death.”
The authors wrote:
This international collaboration — comprised of physicians, like lead author Peter McCullough, MD, courageously treating patients despite the prevalence of “therapeutic nihilism” among government agencies like the NIH and FDA — outlines the urgency of, “prompt early initiation of sequenced multidrug therapy (SMDT) … to stem the tide of hospitalizations and death.”
The authors wrote:
Included in the paper is a “sequential multidrug treatment algorithm” and summaries of the rationale and evidence for each component.The early stage of viral replication provides a therapeutic window of tremendous opportunity to potentially reduce the risk of more severe sequelae in high risk patients. Precious time is squandered with a ‘wait and see’ approach … resulting in unnecessary hospitalization, morbidity, and death. … In newly diagnosed, high-risk, symptomatic patients with COVID-19, SMDT has a reasonable chance of therapeutic gain with an acceptable benefit-to-risk profile.
Dec 8, 2020: Appearing as a witness on Dec. 8, 2020, before the Senate Committee on Homeland Security and Governmental Affairs—which held a hearing on “Early Outpatient Treatment: An Essential Part of a COVID-19 Solution”— Dr. Pierre Kory, President of the Frontline COVID-19 Critical Care Alliance (FLCCC), called for the government to swiftly review the already expansive and still rapidly emerging medical evidence on Ivermectin.
Related Ivermectin and COVID-19 Publications:
- A multi-centre randomised controlled study in Egypt (Elgazzar, Research Square) reported that the death rate was significantly lower in Ivermectin treated patients group (severe patients) vs non-Ivermectin group (2% vs 20%). 1,300 patients were included in this randomized controlled trial.
- This randomized controlled trial out of Iran (Hashim, pre-print) used Ivermectin and Doxycycline in mild, moderate, and severe hospitalized COVID-19 patients. No patients in the mild and moderate COVID-19 category died and 18% of the severe patients perished taking this medication combo. In the control group, no mild-moderate patients died, but 27% of the severe COVID patients died. The patients who also got Ivermectin had a shorter recovery.
- A randomized, double-blind, placebo-controlled, multicenter, phase 2 clinical trial at five hospitals (Iran) and 180 patients with mild to severe disease (Niaee, ResearchSquare, Nov 2020). Ivermectin as an adjunct reduced the rate of mortality, the duration of low oxygen saturation, and the duration of hospitalization.
- The ICON study in US, published in Chest, Oct 2020 reported that Ivermectin treatment was associated with lower death rate vs Control (13.3% vs 24.5%) during treatment of COVID-19, especially in patients with severe pulmonary involvement.
- A double-blinded randomised controlled study in Bangladesh (Mahmud et al) reported that the death rate was 0% (0/183) in the Ivermectin arm vs 1.67% (3/180) in the control arm in mild to moderate COVID-19 patients.
- The IDEA (Ivermectin, Dexamethasone, Enoxaparin and Aspirin) study from Argentina reported 1 death out of 167 patients studied. The patient that died was a severe COVID-19 patient that required ventilator support.
- The pre-AndroCoV trial from Brazil reported that early detection of COVID-19 followed by a pharmaceutical approach with different drug combinations (Azithromycin, Hydroxychloroquine, Nitazonide, Ivermectin) yielded irrefutable differences compared to non-treated controls in terms of clinical outcomes, ethically disallowing placebo-control randomized clinical trials in the early stage of COVID-19 due to the marked improvements.
- A retrospective study out of Bangladesh (Khan, Archivos de Bronconeumologia 2020). This retrospective study enrolled a total of 325 from April to June 2020. 248 adult COVID-19 patients were looked at in two groups, 115 received ivermectin plus standard care (SC), while 133 received only standard care (SC). This study showed that Ivermectin was efficient at rapidly clearing SARS-CoV-2 from nasal swabs (median 4 days). This was much shorter than in the COVID-19 patients receiving only SC (15 days) or receiving a combination of three antiviral drugs (7–12 days). In addition, fewer Ivermectin patients developed respiratory distress leading to ICU admission. In fact, with Ivermectin, there was a quick hospital discharge (median 9 days) in 114 out of 115 patients; the one remaining patient had been admitted with advanced disease.
Ivermectin Contraindications
Asthma: Patients with a history of severe asthma should receive ivermectin with caution. Occasionally, systemic ivermectin has been reported to worsen bronchial asthma.Hepatic disease: Although not extensively studied, due to its extensive hepatic metabolism, ivermectin should be administered with caution in patients with significant hepatic disease.
Human immunodeficiency virus (HIV) infection, immunosuppression: In patients with immunosuppression (including those with human immunodeficiency virus (HIV) infection) treated for intestinal strongyloidiasis, repeated ivermectin courses may be necessary. Adequate and well-controlled clinical studies have not been conducted in such patients to determine the optimal dosing regimen. Several treatments (i.e., at 2 week intervals) may be required and a cure may not be achievable. Control of extra-intestinal strongyloidiasis in these patients is difficult, however, suppressive therapy (i.e., once per month) may be helpful.
Pregnancy: Data with oral ivermectin use during pregnancy are insufficient to inform a drug-associated risk. Systemic exposure from topical use of ivermectin is much lower than from oral use. Four published epidemiology studies evaluated the outcomes of a total of 744 women exposed to oral ivermectin in various stages of pregnancy. In the largest study, 397 women in the second trimester of pregnancy were treated open-label with single doses of ivermectin or ivermectin plus albendazole; there was no observed difference in pregnancy outcomes between treated and untreated populations. However, these studies cannot definitely establish or exclude the absence of drug-associated risk during pregnancy, because either the timing of the administration during gestation was not accurately ascertained or the administration only occurred during the second trimester. In animal embyrofetal development studies of oral ivermectin given during organogenesis, adverse developmental outcomes, including cleft palate, exencephaly, wavy ribs, and clubbed forepaws, occurred at or near doses that were maternally toxic. Pre-implantation loss and abortion were also noted.
Breast-feeding: After oral administration, ivermectin is excreted in human breast milk in low concentrations. Excretion in human breast milk after topical administration has not been evaluated. According to the manufacturer, treatment with oral ivermectin in mothers who are breast-feeding should only be undertaken when the risk of delayed treatment to the mother outweighs the possible risk to the newborn. Previous American Academy of Pediatrics (AAP) recommendations considered oral ivermectin to be usually compatible with breast-feeding. The amount of ivermectin present in human milk after topical application has not been studied; however, systemic exposure from topical ivermectin use is much lower than from oral use. According to the manufacturer, discontinue nursing or discontinue the topical cream, taking into account the importance of the drug to the mother. Women who are breast-feeding while using topical ivermectin should avoid accidental transfer of ivermectin to the breast area where it might be directly ingested while nursing.
Children, infants: The topical administration of ivermectin to infants and children should be under the direct supervision of an adult to prevent ingestion of the lotion.
Onchodermatitis: Patients with hyperreactive onchodermatitis (i.e., sowda) may be more likely than others to experience severe edema and worsening of onchodermatitis after ivermectin use.




.png)

.png)
Comments
Post a Comment